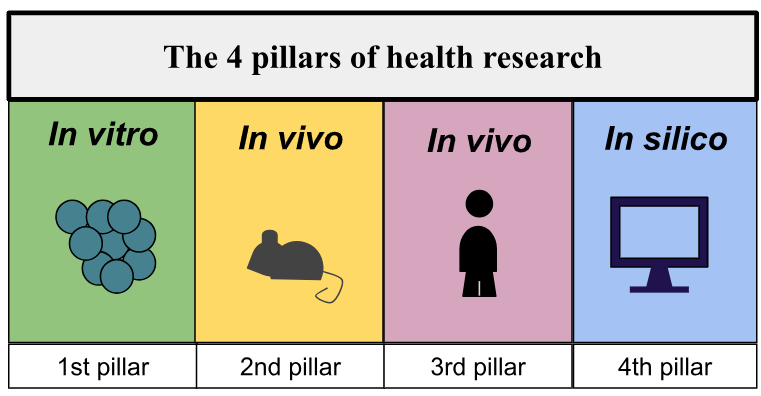
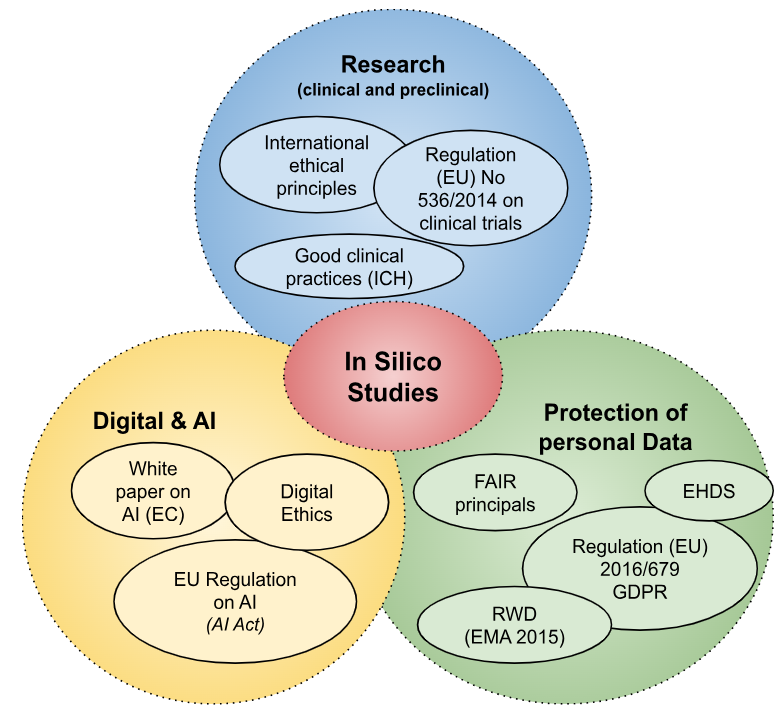
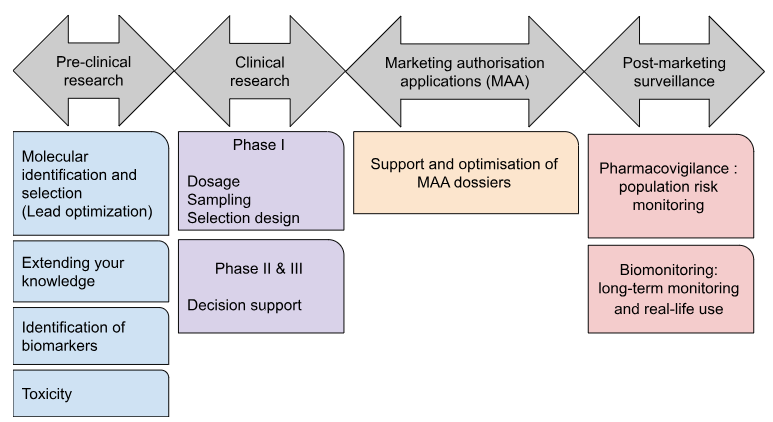
[1] SEBBAN E. and BABINET G., Santé connectée: demain, tous médecins? Une révolution avec les professionnels de santé, pour les patients, ed. Hermann, Paris, 2015, 216p.
[2] CCNE, Report by the National Consultative Ethics Committee, "Numérique et santé: quels enjeux éthiques pour quelles régulations", 19/11/2018, p.6.
[3] F. ROUSSEAU C., CROZATIER C., A. DUVERLE D., BUYSE M., E. CARTER S., M. VOISIN E., BOISSEL F.-H., " A regulatory landscape shift for in silico clinical trials ", Regulatory rapporteur, Vol. 16, N°11, november 2019, pp. 18- 21.
[4] BAJARD A., CHABAUD S., CORNU C. and all, "An in silico approach helped to identify the best experimental design, population, and outcome for future randomized clinical trials", Journal of Clinical Epidemiology, published online 2015.
[5] EDITH, “Building the European Virtual Human Twin”, Deliverable 3.2 First draft of the VHT roadmap, Report, 31 July 2023. (https://zenodo.org/records/8200955).
[6] Avicenna Alliance, "Avicenna Alliance members case studies on in silico medicine", April 2020, p.8: Example of the Extrapolis project. [Available online: https://avicenna-alliance.com/files/user_upload/PDF/Avicenna-Alliance_Members_Case-Studies_04-20.pdf] (Consulted on 07/05/2020).
[7] HUNTER P. J., “The IUPS Physiome Project: a framework for computational physiology”, Progress in Biophysis and Molecular Biology, Vol. 85, Issues 2-3, July 2004, pp.551-569.
[8] GIRARD P., CUCHERAT M., "Simulation des essais cliniques dans le développement des médicaments", Thérapie, May-June 2004, n°3, pp.294.
[9] Leem, "Essais cliniques 2030", survey of French and international experts by IQVIA, 10 March 2022, 97 p.
[10] T. MUSUAMBA F., et al, "Verifying and validating Quantitative Systems Pharmacology and In Silico Models in Drug Development: Current Needs, Gaps, and Challenges", CPT Pharmacometrics & Systems Pharmacology, 2020, 3p.
[11] GIRARD P., CUCHERAT M., "Simulation des essais cliniques dans le développement des médicaments", Op. Cit.
[12] MORRISON Tina, "How simulation can transform regulatory pathways", FDA, 09/08/18, [Available online: https://www.fda.gov/science-research/about-science-research-fda/how-simulation-can-transform-regulatory-pathways].
[13] E-SANTE EUROBIOMED, "Les essais cliniques in silico : le numérique et la modélisation au service du médicament", Rubrique actualité, 29/03/2018, [Available online: https://www.eurobiomed.org/actualite/detail/News/les-essais-cliniques-in-silico-le-numerique-et-la-modelisation-au-service-du-medicament/], (Accessed on 11/04/19).
[14] MANOLIS E., T. MUSUAMBA F., E. KARLSSON K., "Regulatory Considerations for Building an In Silico Clinical Pharmacology Backbone by 2030", Op. Cit.
[15] AFFOLTER C., "Novadiscovery aims for more efficient R&D", AGEFI, 13/03/2020, p.3.
[16] Ibid.
[17] A. SENI D., " L'expérimentation in silico et les nouvelles technologies moléculaires en biopharmaceutique stratégies de R&D des firmes de l'industrie de la région de Montréal ", Gestion, 2003, vol.28, p. 88.
[18] BRIVES C., " L'individu dans un essai thérapeutique. Sur quelques aspects du devenir objet dans les expérimentations scientifiques", Revue d'anthropologie des connaissances, 2013, vol.6, n°3, pp. 653-675.
[19] InSilc, project initiated by the European Commission, official website: https://insilc.eu/, 28 November 2019. European Commission website, https://ec.europa.eu/digital-single-market/en/news/eu-funded-insilc-project-promises-reduce-size-and-duration-human-clinical-trials, 28 November 2019.
[20] M. STOCKES J., YANG K., SWANSON K., S. JAAKKOLA T., BARZILAY R., J. COLLINS J., "A Deep Learning Approach to Antibiotic Discovery", Op. Cit.
[21] BOUZOM F. et al, "Physiologically based pharmacokinetic (PBPK) modelling tools: how to fit with our needs?", Biopharmaceutics & Drug Disposition, 33(2), 2012, p. 55-71
[22] KASTRISSIOS H, GIRARD P., "Protocol deviations and execution models. In: Ko HC", Duffull SB, editors. Simulation for designing clinical trials. New York, USA: Marcel Dekker, 2002
[23] BAJARD A., CHABAUD S., CORNU C. and all, "An in silico approach helped to identify the best experimental design, population, and outcome for future randomized clinical trials", Journal of Clinical Epidemiology, published online 2015
[24] MELLERIN M., " La complexité croissante des études cliniques : Analyse et adaptation des laboratoires pharmaceutiques ", Dir. TABOULET Florence, Thèse pour le diplôme d'état de docteur en pharmacie, Université Toulouse III Paul Sabatier, 8 December 2017, p.80
[25] BOUZOM F. et al, "Physiologically based pharmacokinetic (PBPK) modelling tools: how to fit with our needs?", Op. Cit.
[26] F. ROUSSEAU C., CROZATIER C., A. DUVERLE D., BUYSE M., E. CARTER S., M. VOISIN E., BOISSEL F.-H., "A regulatory landscape shift for in silico clinical trials", Op. Cit.
[27] VEYRAT-FOLLET C., BRUNO R., OLIVARES R. et al. "Clinical trial simulation of docetaxel in patients with cancer as a tool for dosage optimization", Clinical Pharmacology and Therapeutics, n°68, 2000, p.677-687.
[28] HAS, "Evaluation of connected medical devices. Spécificités méthodologiques d'évaluation clinique d'un dispositif médical connecté (DMC)", Service évaluation des dispositifs, 29/01/2019.
[29] https://insilc.eu/about/
[30] NATHAN N., ABOU TAAM R., DELACOURT C., DESCHILDRE A., REIX P. et al, "A national internet-linked based database for pediatric interstitial lung diseases: the French network. Orphanet Journal of Rare Diseases, n°40, 15/06/12.
[31] BAJARD A., CHABAUD S., CORNU C. and all, "An in silico approach helped to identify the best experimental design, population, and outcome for future randomized clinical trials", Op. cit.
[32] EMA, ICH Topic E7, "Studies in support of special populations: Geriatrics. Questions and answers", EMA/CHMP/ICH/604661/2009, European Medicines Agency, London, 2010.
[33] Avicenna Alliance, "Avicenna Alliance members case studies on in silico medicine", April 2020, p.8: Example of the Extrapolis project. [Available online: https://avicenna-alliance.com/files/user_upload/PDF/Avicenna-Alliance_Members_Case-Studies_04-20.pdf] (Consulted on 07/05/2020).
[34] Regulation (EU) No 536/2014 of the European Parliament and of the Council of 16 April 2014 on clinical trials on medicinal products for human use and repealing Directive 2001/20/EC, O.J., L 158, 27 May 2014, Article 10.
[35] MADABUSHI R, BENJAMIN JM, GREWAL R et al, "The US Food and Drug Administration's model-informed drug development paired meeting pilot program: early experience and impact", Clinical Pharmacology & Therapeutics, n°106, 2019, pp. 74-78.
[36] E. MANOLIS, F. T. MUSUAMBA, K. E. KARLSSON, "Regulatory Considerations for Building an In Silico Clinical Pharmacology Backbone by 2030", Clinical Pharmacology & therapeutics, 2020, vol. 107, n°4, pp. 746-748 (747).
[37] https:/whitelabgx.com/
[38] https://www.eu-stands4pm.eu/
[39] BRUNAK S., BJERRE C., CATHERINE E., Ó CATHAOIR K., GOLEBIEWSKI M., KIRSCHNER M., KOCKUM I., MOSER H. AND WALTEMATH D., "Towards standardisation guidelines for in silico approaches in personalised medicine" Journal of Integrative Bioinformatics , vol. 17, no. 2-3, 2020.
[40] https://www.eu-stands4pm.eu/
[41] EMA, « Questions and answers: qualification of digital technology-based methodologies to support approval of medicinal products », Human Medicines Division, EMA/219860/2020, 1/06/2020: https://www.ema.europa.eu/en/documents/other/questions-and-answers-qualification-digital-technology-based-methodologies-support-approval-medicinal-products_en.pdf
[42] EMA, ‘Guideline on quality, non-clinical and clinical requirements for investigational advanced therapy medicinal products in clinical trials’, EMA/CAT/22473/2025, 20 January 2025, p.52.
[43] EMA, ‘EMA Regulatory Science to 2025. Strategic reflection’, December 2018, 60p: https://www.ema.europa.eu/en/documents/regulatory-procedural-guideline/ema-regulatory-science-2025-strategic-reflection_en.pdf
[44] EMA, ‘Future-proofing Qualification of Novel Methodologies (QoNM)’, Action plan, September 2024: https://www.ema.europa.eu/en/documents/other/future-proofing-qualification-novel-methodologies-qonm-action-plan_en.pdf
[45] MANOLIS E, MUSUAMBA FT, KARLSSON KE, ‘Regulatory Considerations for Building an In Silico Clinical Pharmacology Backbone by 2030’, Op. cit, pp. 746-748.
[46] EMA website, https://www.ema.europa.eu/en/committees/working-parties-other-groups/pdco-working-parties-other-groups 21 October 2019.
[47] MARTIS B. S., ROUSSEAU F. C. and all., “The potential of in silico approaches to streamline drug development”, Avicenna Alliance Position Paper, Avril 2023 ; GERIS L. and all., The role of artificial intelligence within in silico medicine, Avicenna Alliance and VPH Institute white paper, August 2022.
[48] EMA, "Guideline on the reporting of physiologically based pharmacokinetic (PBPK) modelling and simulation", EMA/CHMP/458101/2016, 13 December 2018, 16p. [Available online: https://www.ema.europa.eu/en/documents/scientific-guideline/guideline-reporting-physiologically-based-pharmacokinetic-pbpk-modelling-simulation_en.pdf].
[49] EMA, Qualification of novel methodologies for drug development: guidance to applicants’, EMA/CHMP/SAWP/72894/2008, November 2014, 15.p: https://www.ema.europa.eu/en/documents/regulatory-procedural-guideline/qualification-novel-methodologies-drug-development-guidance-applicants_en.pdf
[50] EMA, "Reflection paper on the use of Artificial Intelligence (AI) in the medicinal product lifecycle", EMA/CHMP/CVMP/83833/2023, July 2023, 17p.
[51] EMA, “Reflection paper on the use of Artificial Intelligence (AI) in the medicinal product lifecycle”, EMA/CHMP/CVMP/83833/2023, September 2024.
[52] EMA, “ICH M15 Guideline on general principles for model-informed drug development. Step 5”, EMA/CHMP/ICH/4966426/2024, 9 February 2026.